Haematopoietic stem cell transplantation (HSCT) is one of the most effective treatments for many malig- nant and non-malignant diseases, such as haematological malignancies, some solid tumours, and immunodeficiency diseases1 . In the last decade, there has been a significant increase in the number of patients undergoing HSCT, while the annual number is currently approximately 50,0001 . There are two different types of HSCT: autologous and allogeneic2-4. Although the success rate of HSCT treatment is gradually increasing, the conditioning regimens consisting of intensive chemotherapy and/or radiotherapy given to these patients for 7–10 days before and after transplantation to prevent acute or chronic graft versus host disease cause many side effects5,6. Epileptic seizures, which are the most common side effects and can lead to significant neurological complications, including consciousness-related, motor, and sensory symptoms, affect the success of transplantation7. The incidence of epileptic seizures in patients undergoing HSCT is approximately 5–7%8,9. Seizures that occur during the transplant process are considered a side effect of the drugs used in conditioning regimens. Additionally, studies have shown that these epileptic seizures are related to dose and dura- tion of drug administration10.
In recent years, high success rates have been achieved with the use of melphalan in patients who have undergone autologous transplantation, but the neurotoxic effect of this drug has not been fully determined11. The epileptic seizure complication is generally studied in allogeneic HSCT groups, but similar research on autologous HSCT is quite limited in the literature. When these studies were examined, it was found that epileptic seizures after HSCT were analysed only based on clinical records and that these patients had not undergone a careful semiological and electroencephalographic (EEG) evaluation.
In this study, we aimed to detect EEG anomalies, which may occur after intensive chemotherapy regimens given for 7–10 days before and after transplantation, at an early stage in patients who have undergone allogeneic and autologous HSCT. Additionally, it was our goal to emphasise the importance of EEG monitoring for early diagnosis and management of related non-convulsive seizures that may be life-threatening.
Approval of the Biruni University NonPharmaceutical Clinical Research Ethics Committee was received on 16.02.2022 with the number 2015-KAEK-60-22-07.
Materials and methods
Patients at a private bone marrow transplantation centre from January 2021 to May 2022 who were evaluated and found to be suitable for HSCT treatment were included in the study. Of the 63 patients, 9 patients with a history of epilepsy or psychiatric illness, who were taking anti-seizure or psychiatric medication, had an infection, or experienced electrolyte disorders before and after transplantation were excluded from the study. Additionally, a single patient who died due to drug side effects during the conditioning regimen was omitted from the study. As a result, the study was conducted with 53 patients. The age, gender, educational status, drugs used, HSCT type (allogeneic or autologous), and treatment regimens applied before and after HSCT were recorded. Following the protocol of the transplantation centre as part of the conditioning regimen, drugs such as busulfan, fludarabine, and N90 were started on the day of hospitalization in allogeneic transplantation patients and administered for five days. As anti-seizure therapy for patients undergoing allogeneic transplantation, levetiracetam was started as an oral therapy at a dose of 2x500 mg 12 hours before busulfan. It was continued until 24 hours after the end of the last dose of busulfan. After a five-day preparatory regimen, HSCT was performed on the seventh day of hospitalization. The autologous group was treated with melphalan for two days after admission to the hospital according to the protocol of the transplantation centre, and HSCT was performed on the fourth day of admission. Busulfan was used in only one of the patients who underwent autologous transplantation. As anti-seizure therapy for this patient, levetiracetam was started as an oral therapy at a dose of 2x500 mg 12 hours before the busulfan. In other autologous transplant patients, prophylactic anti-seizure medication was not used since busulfan was not present in the conditioning regimens.
All patients underwent EEG monitoring twice, once on the first day of hospitalization and again one week after the conditioning regimens were started and HSCT was performed. EEG monitoring was undertaken by placing electrodes according to the 10–20 system on the scalp with a 32-channel EEG device in a quiet and dim environment. Hyperventilation and photic stimulation were performed in all patients.
The EEGs were examined and recorded by two neurophysiologists. According to the EEG examination, patients were classified as normal, as having basic activity disorders, as having focal anomalies, and as having generalised anomalies. Patients were then divided into two groups: allogeneic and autologous HSCT patients. The demographic data and pre- and post-transplant EEG characteristics of both groups were compared.
Table 1. Demographic data, educational status, drugs used, and EEG findings of the patients
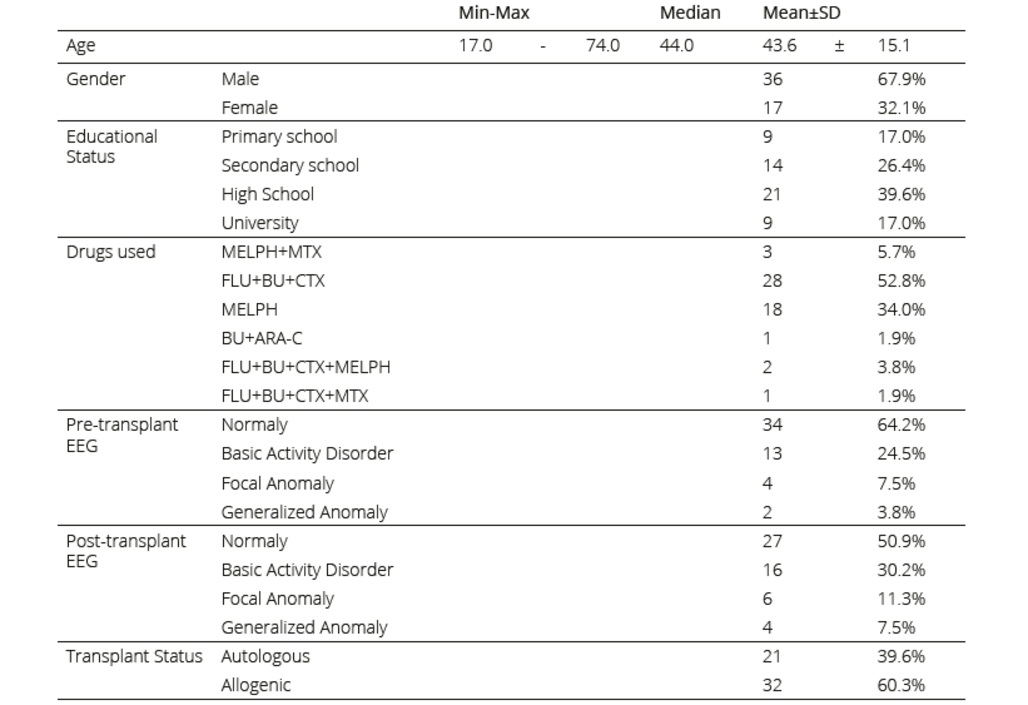 MELPH: melphalan, MTX: mitoxantrone, FLU: fludarabine, BU: busulfan, CTX: cyclophosphamide, ARA-C: cytarabine
MELPH: melphalan, MTX: mitoxantrone, FLU: fludarabine, BU: busulfan, CTX: cyclophosphamide, ARA-C: cytarabine
Statistical method
Mean, standard deviation, median, minimum, maximum, frequency, and ratio values were used in the descriptive statistics of the data. The distribution of the variables was measured by the Kolmogorov–Smirnov test. The Mann– Whitney U test was used in the analysis of the quantitative independent data. In the analysis of the qualitative independent data, the chi-square test was employed, and when chi-square test conditions were not met, the Fisher test was used. McNemar’s test was employed in the analysis of the qualitative dependent data. Finally, the SPSS 28.0 program was used in the analyses. P-values less than 0.05 were considered significant.
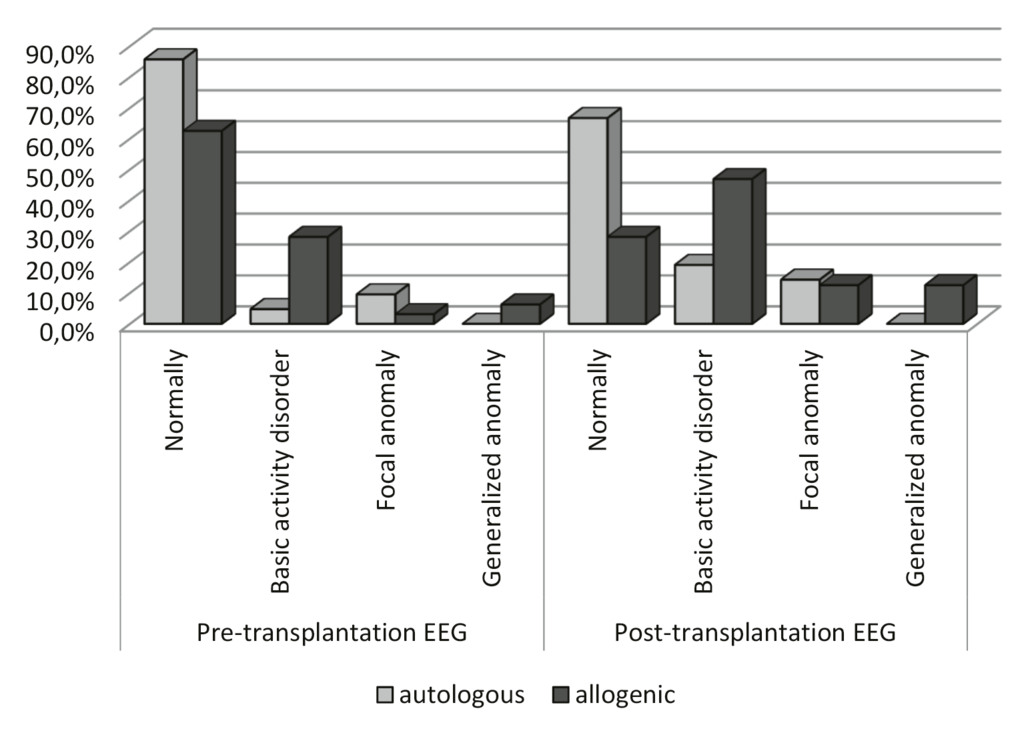
Figure 1. Comparison of EEG findings of the autologous and allogeneic groups
Results
The mean age of the 53 patients who underwent HSCT was 43.6±15.1 years; 17 (32%) were female and 36 (68%) were male. When the pre-transplant EEG findings were examined, 34 (64.2%) patients had normal EEGs, 13 (24.5%) had a basic activity disorder, 4 (7.5%) had a focalanomaly, and 2 (3.8%) had a generalised anomaly. After transplantation, 27 (50.9%) had normal EEG findings, 16 (30.2%) had a basic activity disorder, 6 (11.3%) had a focal anomaly, and 4 (7.5%) had a generalised anomaly. Of the patients, 21 (39.6%) were in the autologous HSCT group and 32 (60.3%) were in the allogenic HSCT group. Demographic data, educational status, drugs used, and EEG findings of the patients are shown in Table 1.
There was no significant (p>0.05) difference between gender and educational status of the patients in the autologous and allogeneic groups. But there was significant difference between the age of the patients in the two groups.
In addition, no significant difference (p>0.05) was found between the autologous and allogeneic groups in the pre-transplant basal EEGs. However, in the allogeneic group, the rate of an anomaly in the post-transplant EEGs was significantly higher than that in the autologous group (p<0.05). While the incidence of EEG anomalies after transplantation increased compared to the incidence before the transplantation in the autologous group, this increase was not statistically significant (p>0.05). There was both a statistically significant increase in post-transplant EEG anomalies (p <0.05) in the allogeneic group and a significant decrease in normal EEGs after transplantation. The most common anomaly in pre-transplant EEGs was a basic activity disorder; the incidence of this type of anomaly was also higher in post- transplant EEGs (Table 2). In the allogeneic group, there was a significant increase in the rates of epileptic anomalies (ground activity disorder, as well as focal and generalised anomalies) detected in EEGs after transplantation compared to before transplantation (Table 2, Figure 1). Busulfan was used as a conditioning regimen in one of the autologous transplantation patients, and while the pre-transplant EEG of this patient was normal, there was a focal anomaly in the post-transplant EEG (Figure 1).
Table 2. Comparison of demographic data, drugs used, and EEG findings of the autologous and allogeneic groups
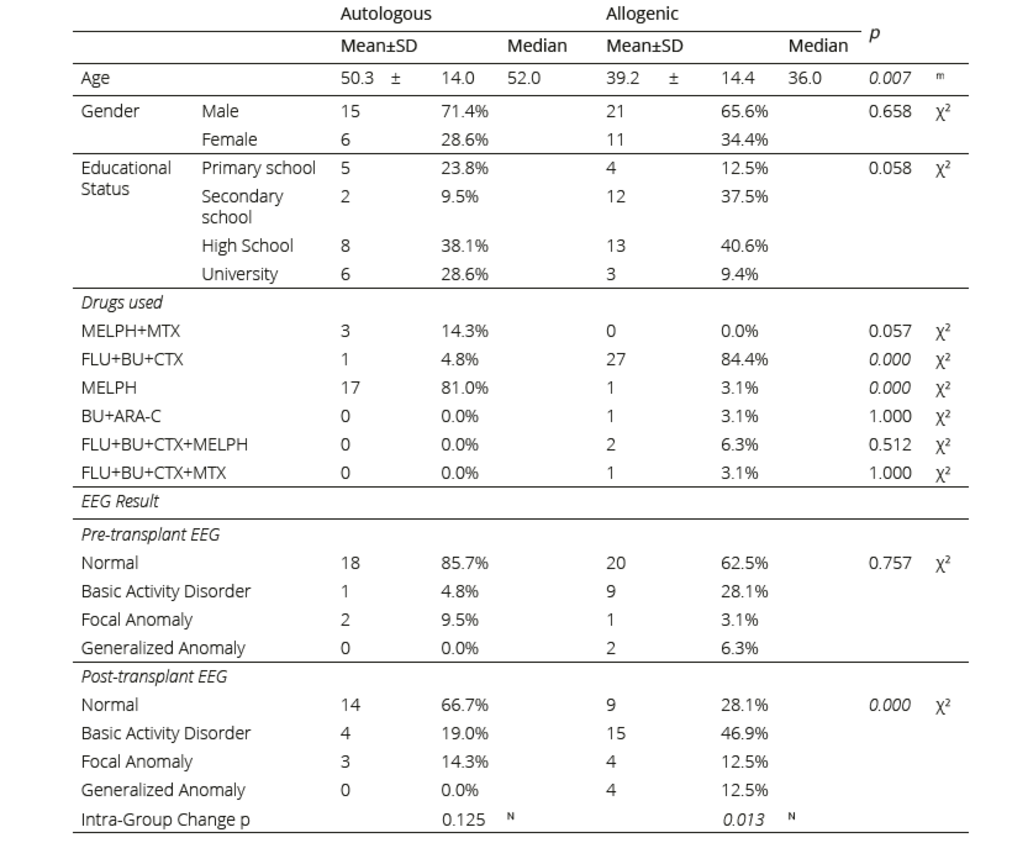
mMann-Whitney u test / χ2: Chi-square test (Fisher test) N: MC: Nemar test
MELPH: melphalan, MTX: mitoxantrone, FLU: fludarabin, BU: busulfan, CTX: cyclophosphamide, ARA-C: cytarabine
Discussion
In this study, the basal EEG monitoring of patients before allogeneic and autologous HSCT and the EEG monitoring performed after HSCT following the appropriate conditioning regimen were evaluated to compare both groups in terms of EEG anomalies. It filled several gaps in the literature as there is no extant study focusing on seizures after allogeneic and autologous HSCT or suggesting a diagnostic and therapeutic approach in such cases. In addition, the epileptic seizure complication is generally evaluated in allogeneic HSCT groups; however, studies on autologous HSCT are quite limited in the literature. Approximately 30–60% of patients un- dergoing HSCT or solid organ transplantation have complications of the central nervous system10. Among these complications, epileptic seizures are common. Zivkovic et al. found the incidence of seizures to be approximately 7–27% in solid organ transplant recipients and approxi- mately 5–7% in HSCT recipients8. In another study, seizures were seen in 10.7% of all patients after HSCT and in 73.6% of patients with neurological complications8,12. In their study of allogeneic HSCT recipients, Zhang et al. reported that intracranial haemorrhage was the most common neurological complication, with 7% of patients having seizures after transplantation and 40% of seizures being associated with drug side effects13. In our study, although epileptic attacks were not observed in our patients, epileptic anomalies, such as a basic activity disorder, as well as focal and generalised anomalies, were observed in the post-transplant EEGs in 33% of the autologous group and 72% of the allogeneic group.
Studies have shown that busulfan used in conditioning regimens in the allogeneic HSCT group may cause epileptic seizures. It has been revealed that the ability of busulfan to lower the seizure threshold develops as a result of its limited binding to plasma protein and its ability to cross the blood–brain barrier (BBB) easily, reaching levels similar to plasma in the CSF, unlike other lipophilic alkylating agents14. Therefore, it is common to apply seizure prophylaxis to the conditioning regimens with this alkylating agent in patients undergoing HSCT15-17. In our study, prophylactic anti-seizure medication was used only in allogeneic group patients when busulfan was part of the conditioning regimen. The autologous group was not given anti-seizure drug prophylaxis. Only a limited number of studies exist on the evaluation of epileptic seizures by EEG monitoring in patients using busulfan in the HSCT conditioning regimen. In these studies, EEG abnormalities were reported in patients who did not have seizures and the neurotoxic effects of the drug were confirmed18,19. In our study, the rate of EEG anomaly after transplantation was higher in the patient group who underwent allogeneic HSCT using busulfan than in those who did not receive this drug. Although EEG anomalies after HSCT are more common in patients treated with busulfan and levetiracetam for prophylaxis, we believe that the latter drug prevents the development of seizures. However, one study suggested that secondary transplantation failure was seen in patients treated with levetiracetam and that this failure may be related to myelodysplastic syndrome due to levetiracetam use20. Therefore, there is a need for studies showing that levetiracetam can be used safely, for epileptic seizure prophylaxis, in patients undergoing HSCT. In this regard, we believe that anti-seizure treatments that do not affect the bone marrow should be used.
While there are many studies on seizures and anti-seizure treatment in patients who have undergone bone marrow transplantation, the number of studies on the autologous group is quite small. Single-agent melphalan at a dose of 200 mg/m² is the international standard for the autologous HSCT conditioning regimenleveti- racetam21. Melphalan, which is administered to autologous HSCT patients, has neurotoxic side effects similar to those of other alkylating drugs that cross the BBB, and these side effects may occur depending on the dose22. In our study, it was found that the melphalan dose range used in seven patients with EEG anomalies after transplantation was 180–200 mg/ m², and the melphalan dose range was lower in patients with normal EEGs. The decrease in the seizure threshold resulting from melphalan neurotoxicity is also due to the combined administration of melphalan with other drugs. In our study, melphalan was applied to one patient in combination, and monotherapy was applied to all other autologous transplant patients. Prophylactic anti-seizure therapy is not used in autologous patients who are prepared for transplantation with melphalan. Studies have reported very low rates of seizures resulting from high- dose melphalan23. In our study, while seizures were not observed in the autologous group patients receiving high doses of melphalan, the rate of EEG anomaly after transplantation was found to have increased compared to that in the pre-transplant period, but the difference was not statistically significant. Although there are no signs and outcomes requiring prophylactic anti-seizure treatment in these patients, the increase in EEG anomaly rate indicates that clinicians need to be more careful. 100.th day policlinic controls, no epileptic seizure or brain activity disorder was observed in any of our patients with EEG anomalies. We think that this situation is achieved with the prophylactic anti-seizure treatment that we started during the preparatory regimen phase. In patients whose EEG findings worsened post transplantation, clinical worsening or seizures were observed in neither groups.
Conclusion
HSCT is a life-saving treatment modality for haematological malignancies, but it has some complications. Epileptic seizures can develop during HSCT that can have a very negative impact on the patient’s prognosis. Therefore, it is important to consider epileptic seizures in the clinical follow-up of HSCT patients, and the crucial nature of EEG monitoring should be noted for early diagnosis and treatment of non-convulsive clinical manifestations. In the evaluation of impaired consciousness seen in HSCT patients, first of all, it should be decided whether there is a seizure by analysing the EEG results. Then, the causes and risk factors of the seizure should be determined, and the relevant neurological, laboratory, and imaging examinations should be performed as soon as possible. Although the higher rate of EEG anomalies in the allogeneic group supports the known side effects of busulfan, we believe that high doses of melphalan used in the autologous group may also have caused EEG anomalies; therefore, further research is necessary to support this finding.



 MELPH: melphalan, MTX: mitoxantrone, FLU: fludarabine, BU: busulfan, CTX: cyclophosphamide, ARA-C: cytarabine
MELPH: melphalan, MTX: mitoxantrone, FLU: fludarabine, BU: busulfan, CTX: cyclophosphamide, ARA-C: cytarabine


COMMENTS
0 comments